Abstract
Background:
Down syndrome is known for its leukemia predisposition effects. Children with Down syndrome myeloid leukemia who fail chemotherapy, have a poor outcome. No targeted therapies are available for this population. We have generated and extensively characterized several patient-derived xenograft (PDX) models of Down syndrome AML (Barwe et al., 134:2683, Blood, 2019). We evaluated the efficacy of a combination of DNA hypomethylating agent, azacitidine and Bcl2 inhibitor, venetoclax in PDX model of Down syndrome AML. Although this drug combination is used in the clinic for the treatment of adult AML, their combinatorial mechanism of action is not well known.
Methods:
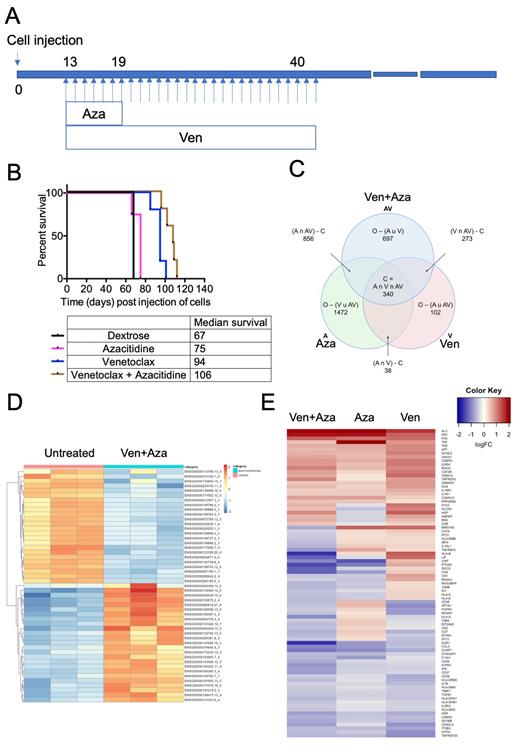
NTPL-386 (3 x 10 6 cells) were injected intravenously in NSG-SGM3 mice. Disease progression was monitored by determination of the percentage of human chimerism in peripheral blood by flow cytometry. Mice were randomly assigned to treatment groups (n=5) on day 13 when human CD45+ cells were detectable in blood. Azacitidine (2.5 mg/Kg) was administered intraperitoneally on days 13-19 and venetoclax (100 mg/Kg) was delivered orally on days 13-40 (Fig. 1A). Mice were monitored daily for pre-determined experimental endpoints and were euthanized when any of the endpoints were attained. Kaplan-Meier survival plots were generated. AML cells were harvested from treated mice and lysed, RNA was isolated and sequenced. Pathway analysis was performed to identify the mechanism of action of these two drugs in combination.
Results:
Azacitidine treatment for one week increased median survival by 8 days compared to untreated mice (Fig. 1B). Venetoclax treatment for 4 weeks was more effective than azacitidine and extended survival by 27 days (P<0.005, compared to untreated). We observed that the combination of venetoclax and azacitidine was much more efficient and prolonged survival by 39 days in this PDX model of refractory Down syndrome AML (P<0.005 and P<0.05 compared to single agent azacitidine or venetoclax respectively). This combination performed better than chemotherapy consisting of cytarabine and daunorubicin, which showed a 11-day improvement in median survival compared to untreated in our previous study (Barwe et al., 134:2683, Blood, 2019).
To understand the mechanism of synergism between these two drugs, we conducted transcriptome analysis on cells harvested from mice (n=3 per group) receiving venetoclax and azacitidine, alone or in combination. The log fold changes for each treatment with respect to untreated were calculated and filtered based on FDR>0.05. The genes that were differentially regulated in the combination treatment (Fig. 1C, blue circle in the Venn diagram) were subjected to ToppGene analysis. A heatmap of the differentially regulated genes shows clustering of the replicates from individual mice (Fig. 1D). 'Hematopoietic cell lineage' and 'cytokine signaling in immune system' were the top modulated pathways in the combination treatment. A heatmap of the logFC of the differentially regulated genes from this pathway showed the modulation of these genes in the combination in comparison with individual drug treatment (Fig. 1E). The differentially regulated genes in the combination treatment that showed high variance from the range defined by individual drug treatment belonged to one of three categories - cell surface proteins, cytokines, and stem cell markers. These data indicate that the synergistic downregulation of cytokine signaling is likely responsible for the combinatorial effect of venetoclax and azacitidine.
Conclusion:
The combination of venetoclax and azacitidine was effective in prolonging survival in a highly refractory Down syndrome AML PDX model (NTPL-386) generated in the laboratory. The top genes differentially regulated by the combination treatment which varied the most in magnitude from the individual drug treatment identified genes that are likely to contribute to the synergism between the two drugs.
Barwe: Prelude Therapeutics: Research Funding. Gopalakrishnapillai: Geron: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal